A research team has found that when the bacteria Klebsiella pneumoniae become resistant to chlorhexidine it can also grant them resistance to colistin, a ‘last-line’ antibiotic. The potential for an organism to become resistant to an antibiotic as a result of its resistance against a biocide has been debated in the past, but this is one of the first instances where there’s been evidence of it happening.
Chlorhexidine is an antiseptic and a common ingredient in many household items such as toothpaste, mouthwash and antiseptic creams and powders. It’s also widely used in hospitals, in products such as hand washes and antiseptic wipes and is often a crucial part of surgery as a way to prevent infection. Colistin is an antibiotic which is classified as a last-resort. That’s because these antibiotics tend only to be used after all other possible drugs have failed to do anything, showing activity against resistant organisms. Colistin works by attaching to the cell membrane of bacteria, by binding to structures called lipopolysaccharides (LPSs) and phospholipids. It then disrupts the membrane, causing it to break apart and letting all the important parts of the cell leak out – leading to the death of the bacteria.
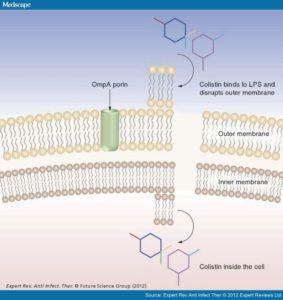
Dr. Matthew E. Ward and his team published their findings in the journal ‘Antimicrobial Agents and Chemotherapy’ on the 31st October. Previously, they had been able to show that K. pneumoniae can become resistant to chlorhexidine under increasing concentrations.
The team first found that when colistin was used against a range of K. pneumoniae strains, some of which were already resistant to the action of chlorhexidine and some which were susceptible to the antiseptic (wild-type), five out of the six chlorhexidine-adapted strains became much more resistant to colistin. The concentration of the antibiotic needed to stop their growth jumped up from 2-4 mg/L to over 64 mg/L.
Afterwards, they added two different efflux inhibitors, CCCP and PaβN, to the chlorhexidine-adapted and wild-type K. pneumoniae strains and treated the strains with chlorhexidine, to see what effect this would have on their growth. Efflux pumps are a major way that bacteria can show resistance to antibiotics and other compounds which can kill them or inhibit their growth – if any of these chemicals manage to enter the bacterial cell, efflux pumps let the bacteria shoot them back out before they can do any damage. Like the name suggests, efflux pump inhibitors stop the pumps from doing their job – so if the bacteria relies on them to escape the killing effect of an antibiotic, the inhibitors would put the organism right back in the drug’s firing line. What they found was that when the strains had the CCCP inhibitor, both the wild-type and the chlorhexidine-adapted K. pneumoniae became a lot more susceptible to chlorhexidine. This strongly suggests that their resistance towards chlorhexidine comes from efflux pumps.
By analysing the genome of the strains, the team were able to pinpoint genes where mutations could give them resistance to chlorhexidine and colistin – smvR and PhoPQ. All of the strains that showed resistance to chlorhexidine had mutations in these genes.
smvR controls how much an efflux pump called smvA is expressed – when the gene was deleted or altered through mutation, this control went out the window and the number of smvA pumps rose, which in theory would let more chlorhexidine be removed from the cell.
Mutations to the PhoPQ gene have been previously linked with resistance to colistin, probably by boosting the activity of pmrK – a protein which can alter the bacterial cell’s outer structure by making the membrane more positively charged. When this happens, it means that colistin finds it harder to bind, and so it can’t really do anything to the bacteria.
According to the authors of the paper, the mutation in PhoPQ which gives resistance to colistin probably comes about as a result of K. pneumoniae developing resistance to chlorhexidine – but they also concluded that there’s a good chance more than one factor can cause the bacteria to become adapted to chlorhexidine aside from the smvR mutation.
The conclusion of the study was that K. pneumoniae can become resistant to colistin, a last-line antibiotic, as a by-product of the bacteria becoming resistant to chlorhexidine, an antiseptic often used in household and hospital disinfectants. Co-author of the paper, Dr. J. Mark Sutton, commented on the implications chlorhexidine resistance could have on our healthcare system in a press release: “Chlorhexidine is a critical part of current infection control practises, and the development of increased resistance to this compound has potential implications for our ability to prevent infections during routine and emergency surgery, and during admission to hospitals.”

Of course, the fact that use of a major infection preventative – especially in hospitals where such precautions are vital to patient healthcare – can lead to the development of resistance against a potentially life-saving antibiotic is a massive concern. But the fact that we’re starting to narrow down the potential mechanisms by which chlorhexidine / colistin cross-resistance happens is a good sign that we’re moving in the right direction to deal with this particular threat.